Cost-Effectiveness of Ultrasound-Guided Liver Biopsy
HEPATOLOGY, May 1998, p. 1220-1226, Vol. 27, No. 5
Original Articles
Cost-Effectiveness of Ultrasound-Guided Liver Biopsy
Tousif Pasha1, Sherine Gabriel2,3, Terry Therneau3, E. Rolland Dickson1, and Keith D. Lindor1
From the 1 Division of Gastroenterology, 2 Department of Internal Medicine, and3 Department of Health Sciences Research, Mayo Clinic, Rochester, MN
ABSTRACT
The risk of a major complication from “blind” percutaneous liver biopsy is reported to be in the range of 0.24% to 3.8%. In a recent randomized trial, patients whose liver biopsies were performed with ultrasonography had a significant reduction in complications requiring hospitalization compared with patients without ultrasound-guided biopsies (0.5% vs. 2.2%, P < .05). Despite this, routine use of ultrasonography for liver biopsies has not been implemented because of controversies with respect to cost-effectiveness. The aim of our study was to analyze the relative cost-effectiveness of performing ultrasound-guided liver biopsies using decision analysis. A decision tree was constructed to compare a strategy of liver biopsy using ultrasonography with a strategy without ultrasonography. The major outcomes included were minor complications such as pain requiring analgesics and major complications, which require hospitalization. Costs included were direct medical costs from the payer’s perspective. In our baseline model, the cost from complications per patient with and without ultrasonography was $62 and $129, respectively. The marginal effectiveness expressed as the number of major complications avoided was 1.2/100 liver biopsies. The incremental cost to avoid one major complication was $2,731. The model was most sensitive to the frequency of major complications and the additional cost of ultrasonography. Our decision analysis model suggests that ultrasound-guided liver biopsy is cost-effective. Future studies assessing the efficacy of image-guided liver biopsies should be conducted.(HEPATOLOGY 1998;27:1220-1226.)
INTRODUCTION
Despite advances in noninvasive imaging procedures and laboratory tests, histological examination of a liver specimen remains essential for the diagnosis of most liver diseases. In addition, histological examination of the liver is extremely valuable to assess the prognosis and to monitor the effects of therapy on the liver. There are several techniques by which a liver specimen can be obtained, and the choice of the technique is dictated by several parameters such as associated coagulation disorders, the presence of ascites, and the results of previous ultrasound examinations.
Percutaneous outpatient liver biopsies are being performed frequently as a result of major advances in the therapy of liver diseases. Although the safety and cost savings of outpatient liver biopsies have been established,1-4 the procedure must not be considered trivial. Complications can range from minor, such as pain at the biopsy site or skin hematoma, to major, such as hemorrhage, hypotension, perforation, hemothorax, and pneumothorax, which require hospitalization. Several modifications of the technique have been suggested to lower the incidence of complications, such as the use of smaller-caliber needles and the use of image guidance.5-8 Ultrasonography aids in directing the needle away from large blood vessels, bile ducts, gallbladder, and colon, and thus potentially reduces complications. However, routine use of image guidance for liver biopsies has not gained widespread acceptance because of controversies with respect to the efficacy and costs. In a recent large multicenter randomized controlled trial, patients who underwent liver biopsies with ultrasound guidance had a significant reduction in complications from liver biopsy requiring hospitalization compared with patients without ultrasound-guided biopsies (0.5% vs. 2.2%, P < .05).8 In addition, minor complications such as pain following biopsy requiring treatment (narcotic and/or nonnarcotic analgesics) were also lower in the group who had ultrasonography (37% vs. 50%, P = 0.003). The authors concluded that, although the use of ultrasonography for guidance of percutaneous liver biopsy led to a significant reduction in the rate of hospitalization, further studies addressing the economic impact of this recommendation are required.
PATIENTS AND METHODS
The aim of our study was to analyze the relative cost-effectiveness of performing ultrasound-guided liver biopsies. To answer this question, we used decision analysis.
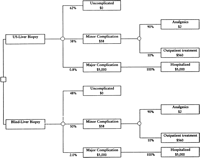
Decision Tree. We constructed a decision tree to compare a strategy of liver biopsy with ultrasonography versus a strategy without ultrasonography (Fig. 1). The first two branches from the decision tree that originate from the square decision node represent the choice between ultrasound-guided liver biopsy and “blind” liver biopsy. Each subsequent branch emanating from a round chance (probability) node reflects the possible outcomes that may occur following liver biopsy. A patient may either have an uncomplicated course or develop complications from the biopsy. The complications considered in our model were minor, such as pain at the biopsy site requiring analgesics or outpatient treatment, and major, such as hemorrhage, hypotension, perforation, hemothorax, and pneumothorax, which require hospitalization. Each event occurs with a given probability and results in monetary costs (Fig. 1).
|
|
Fig. 1. Decision tree used to evaluate the cost-effectiveness of ultrasound-guided liver biopsies. |
The baseline estimates for the analysis were based on data from a randomized study comparing blind and ultrasound-guided liver biopsies.8 Other data for this analysis were obtained from a MEDLINE search of published literature and from reviewing the references of published articles (Tables 1 and 2). To assess the economic effects and potential benefits of ultrasonography in obtaining liver biopsies, we chose figures so as to be consistently biased in favor of “blind” percutaneous liver biopsies. The final outcome measure in the model estimates the additional cost to prevent one major complication, expressed as incremental cost per major complication prevented.
| View This table | table 1. Complication Rates of “Blind” Liver Biopsy |
| View This table | table 2. Summary of Outcomes in Studies Using Ultrasound-Guided Outpatient Liver Biopsies |
Patients and Procedure. The patient population considered in our analysis is composed of those patients who would be considered for an outpatient percutaneous liver biopsy. This procedure is done by trained personnel using standard techniques in patients suspected of having diffuse parenchymal liver disease with normal or near normal coagulation parameters, in the absence of ascites or extrahepatic biliary obstruction. The usual practice following the procedure is to observe the patient for an additional 3 to 4 hours for development of complications such as hypotension, bleeding, or pain.1-4 In the absence of these, the patient is discharged with instructions to stay within 30 minutes from a hospital for a period of 24 hours. In our model, patients would undergo either a “blind” percutaneous biopsy or ultrasound-guided liver biopsy. The type of biopsy needle used and the quality and quantity of specimens obtained by the two strategies were assumed to be identical. The only difference between the two strategies would be the difference in the clinical outcomes (complication) and the cost of the procedure.
Clinical Outcomes. The clinical outcomes considered in the model are shown in Fig. 1. The branches emanating from a round node (chance node) represent whether a patient develops complications, and if so, whether the complications are minor or major. The proportion of patients experiencing each outcome can be calculated by multiplying the probabilities assigned to each chance node along the path leading to that outcome. To calculate the average cost per patient for each strategy, we combine the data on the probability of a major complication with the estimated cost of treating that complication and add the cost of the procedure.
Minor Complications. These usually include pain at the biopsy site or right shoulder requiring analgesics. Pain following liver biopsy may indicate a small amount of blood or bile in the right hypochondrium or on the undersurface of the diaphragm; this has been reported variously in 5% to 50% of patients. 4,8,9Lindor et al. reported a lower frequency of pain after liver biopsies performed with ultrasonography (37.4% vs. 50.1%, P = .003).8 To determine the duration of pain and resources utilized for this complication, we used original data available to us from that study. The data forms of a random sample of 50 patients who were classified as having minor complications were evaluated to determine the duration of pain and treatment sought. Based on this information, we estimated an average duration of pain of 24 hours and that acetaminophen was the analgesic most often used. Only a minority of patients sought care from their physician for this complication (2/50). In our baseline analysis, we assumed that 90% (range, 80%-100%) of patients who develop minor complications would be treated with over-the-counter analgesics (retail cost $2) and the remainder would require outpatient medical care. The cost of an episode of outpatient care was calculated using institutional charge data (see Appendix ).
Major Complications. In our study, a major complication was defined as any complication that required hospitalization. Mortality was not included as an outcome because there is no data showing any significant mortality difference between patients who have image-guided biopsies and those who have “blind” biopsies. Complication rates from previous studies in which patients underwent liver biopsies in the inpatient setting are lower (0.1% to 0.6%) than those found in more recent studies reporting complication rates from outpatient procedures (table 1). In a survey of the literature comprising more than 10,000 inpatient liver biopsies, Terry10 found a mortality rate of 0.12% and an incidence of major complications of 0.32% (table 1). Piccinino et al., in a multicenter study of 68,276 liver biopsies reported a mortality rate of 0.009% and an overall complication rate of 0.22%.11 On the other hand, the average major complication rate was higher in studies using data from outpatient liver biopsies (2.8%). The variability in the major complications reported in the inpatient and outpatient series are probably related to the differences in the severity of complications reported. Whereas 32 of 369 complications (8.7%) reported in the inpatient series10-14 required surgical intervention and 50 of 516 (9.7%) resulted in death, only 2 of 82 complications (2.4%) reported in the outpatient series 1-4,15-19 required surgical intervention and none resulted in death. Furthermore, in studies that have used hospitalization as their definition of major complication, the reported incidence of complications in patients with percutaneous “blind” liver biopsy has been higher (2.2%-5.4%).2,4,8 The average length of hospitalization after admission for a major complication from liver biopsy is reported to be in the range of 1.5 to 3.5 days.2,4,8 Of the 44 hospitalized patients reported by Perrault et al., 39 were discharged within 36 hours of admission and the remaining 5 within 4 days.2Janes and Lindor reported an average hospital stay of 1.5 days.4 The average length of stay reported by Lindor et al. was 3.5 days, and the average hospital charges were $4,066.8 In our analysis, cost of hospitalization for a major complication was $5,000, with a range from $1,000 to $10,000 (see Appendix ).
Effectiveness. Imaging with ultrasound allows direct visualization of the projected needle trajectory. It helps direct the biopsy away from major vessels, bile ducts, gallbladder, and colon, and excludes the presence of anatomic variations or lesions, such as hemangiomas, that may interfere with safe performance of the procedure. Visualization may allow for the selection of an optimal needle path and may be the explanation for the lower complication rate seen with image-guided procedures compared with liver biopsies obtained “blindly.” In addition, ultrasound guidance reduces the number of passes necessary to obtain tissue, which has been shown to correlate with a higher complication rate.2 Lindor et al., in a controlled study (n = 836) comparing liver biopsy with ultrasonography (n = 423) and without (n = 413), reported a major complication rate of 0.5% in the group with ultrasonography compared to 2.2% in the group without ultrasound guidance (P = 0.04).8 All patients who had a major complication were hospitalized, and the mean length of stay was 3.5 days. In an Italian study5 in which a part of the study population was randomized to ultrasound-guided anterior subcostal liver biopsy (n = 170) and conventional Menghini liver biopsy (n = 170), the complications were significantly reduced (0.59% vs. 4%) in the ultrasonography group (table 2). Adjusting for the difference in the definition of major complication used in this study lowers the complication rate from 4% to 2.35%; despite this, the use of ultrasonography reduced the major complication rate by 75%. In an uncontrolled study, Colombo et al. reported a 0.6% major complication rate in 1,192 consecutive ultrasound-guided liver biopsies.9 Smith et al. reported the safety of outpatient ultrasound-guided liver biopsy in 250 biopsies.7 The complication rate defined as unplanned admissions was 1.7% in this study (3/175). Thus, there are emerging data to support the hypothesis that image-guided percutaneous liver biopsies have a lower complication rate than do “blind” liver biopsies. The percentage of major complications reduced by ultrasonography in Lindor’s study was 78% versus 75% in the Italian study. In our baseline analysis, we used 60% reduction in major complications with the use of ultrasonography and a range from 45% reduction to 75% reduction in the sensitivity analysis (table 3).
| View This table | table 3. Probabilities and Cost Estimates Used in the Baseline Analysis and Range for Sensitivity Analysis |
Cost Estimates. The perspective of the analysis was that of a payer or an insurance company. We considered the direct medical costs of each strategy, which include the costs of liver biopsy, biopsy complications, and treatment. To avoid institutional or regional bias in cost estimates, we used costs based on Diagnosis Related Groups reimbursement data and Medicare charge data (Tables 3 and 4). Although costs of care vary among insurers, the Medicare fee schedule is increasingly becoming the basis for computing medical care reimbursement for many indemnity and managed care insurance plans. Liver biopsy costs were determined from current procedural terminology codes (table 4). The average cost of analgesics (primarily over-the-counter drugs from a pharmacy survey) was estimated at $2. The average cost of outpatient management of minor complications was estimated from institutional charges (see Appendix ). Indirect costs, such as travel expenses, lost wages, and other opportunity costs were not included in the model. Our analysis did not account for the intangible cost of the pain and suffering incurred by the patients.
| View This table | table 4. Results Per 1,000 Patients |
Determination of Cost-Effectiveness. The effectiveness of ultrasound-guided liver biopsy was compared with the effectiveness of “blind” percutaneous liver biopsy by determining the reduction in major complications in patients from using this strategy. The cost-effectiveness ratio was calculated by dividing the incremental (marginal) costs in dollars (net costs from liver biopsy performed with and without ultrasonography) by the incremental effectiveness (measured as major complications prevented) between the two strategies. This ratio is expressed as the cost to prevent a major complication, in dollars.
Sensitivity Analysis. To determine which variables in the model had the greatest impact on our results, we performed sensitivity analysis by systematically varying the baseline assumptions and costs over a plausible range of values. The aim of this analysis was to determine threshold values for variables resulting in cost savings as well as to determine threshold values that would yield an incremental cost of preventing a major complication of $10,000 or less. Most authorities agree that if an intervention costs more than $100,000 to save 1 year of life, it is not cost-effective. However, there is no accepted upper limit to comparing the cost-effectiveness of interventions that reduce morbidity. In our analysis, we arbitrarily chose a value of $10,000. Hence, in our analysis, if the cost to prevent a major complication was less than $10,000, we considered it a favorable scenario. As changes in several variables can occur simultaneously, we also reported our results as the best-, intermediate-, and worst-case scenarios from the payer’s perspective (table 5). We expressed effectiveness of ultrasonography as its ability to prevent a major complication. Because the basis of our model depends on the hypothesis that ultrasonography decreases the complication rate after liver biopsy, we varied the frequency of major complications for each strategy (with and without ultrasonography) over a range of literature values and recalculated our results to assess how sensitive our conclusions were to specific components of the model. In addition, we also varied the proportion of major complications (percent reduction in major complications or efficacy of ultrasonography) associated with the use of ultrasonography to determine its impact on our conclusions.
| table 5. Results of the Decision Analysis and Sensitivity Analysis: Best-, Intermediate- (Baseline), and Worst-Case Scenarios from Payer’s Perspective for Ultrasound-Guided Liver Biopsy |
RESULTS
In our baseline model, the average cost from complications per patient undergoing liver biopsy with and without ultrasonography was $62 and $129, respectively (Fig. 1). The difference between the two strategies, or the incremental cost, was $67. Using the additional cost of ultrasonography of $100, the average costs per patient were $534 and $501 for liver biopsies with and without ultrasonography, respectively. This result is extremely sensitive to the cost estimate of the procedures. The incremental cost per patient, which depends on the additional cost of ultrasonography, is a better measure, as it does not depend on the cost estimate of the procedure. The marginal effectiveness in our baseline analysis, expressed as the number of major complications prevented by using ultrasonography, was 1.2 per 100 liver biopsies. The incremental (marginal) cost per major complication prevented in the baseline (intermediate) analysis was $2,731. The model was most sensitive to the frequency of major complications and the additional cost of ultrasonography. Results per 1,000 patients are shown in table 4. table 5 shows the results of the sensitivity analysis as the best-, intermediate- (baseline analysis), and worst-case scenarios from the payer’s perspective, based on varying several variables simultaneously.
In the study by Lindor et al.,8 the efficacy of ultrasonography was 75%; that is, 1.7 complications would be avoided per 100 biopsies if ultrasonography were used. Using these parameters and assuming the additional cost of ultrasonography of $100, the incremental cost to prevent one major complication with the use of ultrasonography would be $406.
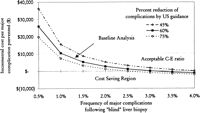
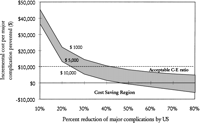
Sensitivity Analysis. Frequency of Major Complications and Percent Reduction of Major Complications. Keeping all variables in our baseline analysis constant, the model was tested for its stability using the range of major complications reported in the literature and the percent reduction in major complications by ultrasonography. The results of this analysis are shown in Fig. 2. In our analysis, the cost of complications with ultrasonography was $62, with a range of $27 to $97, in the worst- and best-case scenarios, and the cost without ultrasound was $129 (range, $39-$329; table 5). Using a baseline cost of ultrasonography of $100, the incremental cost to prevent a major complication was less than $10,000 if the frequency of major complications following “blind” liver biopsies was greater than 1% and if ultrasound guidance reduced major complications by more than 60% (Fig. 2). The model predicts potential cost savings (assuming the additional cost of ultrasonography = $100) if the frequency of major complications was greater than 2.5% and ultrasonography reduced major complications by more than 75% (Fig. 2). However, if the major complication rate of “blind” liver biopsy was less than 1%, the cost-effectiveness ratio (C-E ratio) becomes increasingly unfavorable even if ultrasonography reduces the occurrence of most of the major complications.
|
|
Fig. 2. Two-way sensitivity analysis: incremental cost per major complication prevented as a function of frequency of major complications following “blind” liver biopsy and percent reduction of complications by ultrasonography. |
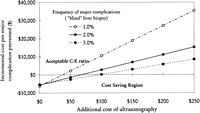
Cost of Procedure. At the frequency of major complications used in our baseline analysis (2%, with a 60% reduction by ultrasonography), the additional cost of ultrasonography that would result in a favorable C-E ratio (<$10,000) was ![]() $187 (Fig. 3) and the cost of ultrasonography that would equalize the average cost of major complications (~$5,000) was $127. The threshold value of ultrasonography below which potential cost savings could be expected was $67 in the baseline analysis, and the range was $12 in the worst-case scenario and $232 in the best-case scenario (table 5). The additional cost of ultrasonography below which potential cost savings could be expected is also shown in Fig. 4. Using data from Lindor et al. (2.2% major complication rate with a 78% reduction by using ultrasonography),8 the cost of ultrasonography below which cost savings can be expected was estimated to be $92.
$187 (Fig. 3) and the cost of ultrasonography that would equalize the average cost of major complications (~$5,000) was $127. The threshold value of ultrasonography below which potential cost savings could be expected was $67 in the baseline analysis, and the range was $12 in the worst-case scenario and $232 in the best-case scenario (table 5). The additional cost of ultrasonography below which potential cost savings could be expected is also shown in Fig. 4. Using data from Lindor et al. (2.2% major complication rate with a 78% reduction by using ultrasonography),8 the cost of ultrasonography below which cost savings can be expected was estimated to be $92.
|
|
Fig. 3. Two-way sensitivity analysis: incremental cost per major complication prevented as a function of frequency of major complications following “blind” liver biopsy and additional cost of ultrasonography. |
|
|
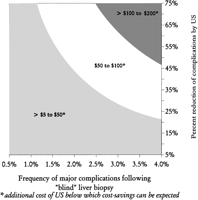
Fig. 4. Three-way sensitivity analysis: frequency of major complications following “blind” liver biopsy, percent reduction of complications by ultrasonography, and additional cost of ultrasonography (for example, if the expected complication rate from “blind” biopsies was 2% and ultrasonography reduces the complication rate by 50%, cost savings can be expected if the cost of ultrasonography is between $50 and $100). |
Cost of Complications. The cost of treating a major complication used in our analysis was direct costs from hospitalization. Using a range of $1,000 to $10,000 did not change the conclusions of our analysis (Fig. 5). Similarly, the frequency or cost of minor complications in the range used did not have a significant effect on the incremental C-E ratio.
|
|
Fig. 5. Two-way sensitivity analysis: incremental cost per major complication prevented as a function of cost of major complication and percent reduction of complications by ultrasonography. |
DISCUSSION
If ultrasound-guided liver biopsy did not increase costs, it would be widely accepted, even if it were for the simple fact that it relieves some of the anxiety and apprehension associated with “blind” percutaneous biopsies. Data from two controlled studies support the hypothesis that ultrasound-guided liver biopsies are associated with lower hospitalization rates from major complications and thus may potentially decrease morbidity and reduce costs. 5,8 In our study, decision analysis was used to compare the cost-effectiveness of liver biopsies with and without ultrasonography.
Spurred by the rapid increase in health care expenses, cost-effectiveness analyses are increasingly being advocated as techniques for comparing the negative (costs) and positive (benefits, effectiveness) aspects of alternative uses of resources to optimize delivery of health care. These techniques have several limitations and should not be the only basis for a decision; however, they can aid in making decisions, give structure to the problem, allow consideration of relevant effects of a decision and also identify key assumptions. Thus, one can use decision-analytic methods and sensitivity analysis to address issues that may not have obvious answers in the form of data from controlled studies. In addition, these analyses may help identify areas in which our knowledge may be deficient and thus provide direction for future investigations.
In our baseline model, assuming that ultrasonography decreases the incidence of complications from percutaneous liver biopsies by 60%, ultrasonography would have a C-E ratio similar to that of other medical interventions. The cost to prevent a major complication was $2,731, which is close to the current cost of a variety of health care procedures; for example, the incremental cost to prevent a severe adverse reaction using low-osmolality contrast medium for intravenous urography was estimated at $17,974.20 If the additional cost for ultrasonography decreases or the cost of managing major complication increases, adopting a strategy of image-guided liver biopsies could provide cost savings. Because the frequency of major complications is quite low in both populations, the incremental C-E ratio closely corresponds to the additional cost of ultrasonography; any additional increase in the procedure cost would result in a significant decrease in the marginal C-E ratio.
Complication rates reported in the literature are variable (table 1). The definition used to classify a major complication may be one reason for this variability. We used hospitalization as the criterion to define a major complication because this outcome can be quantified. In addition, this entails a definite change in management with necessary resource utilization, and for the most part this outcome is associated with a decline in the quality of life. As mentioned earlier, mortality was not included as an outcome because there are no data showing any significant mortality difference between image-guided biopsies and “blind” biopsies. Hence, we could not assign death as an outcome measure in either group without additional assumptions.
Sensitivity analysis confirmed that our model was robust and supported our conclusions over a wide range of values. The most significant determinants of the analysis were the frequency of major complications in the two groups and the additional cost of the ultrasonography. The frequency of major complications (defined as hospitalization) after “blind” percutaneous liver biopsies reported in the literature is consistently greater than 1.5% to 2%. The efficacy of ultrasound-guided liver biopsy can vary from one institution to another. This limitation was evaluated in our study, using sensitivity analysis, by varying the percent reduction in complications with ultrasonography (efficacy of ultrasonography) compared with complications with blind biopsy. This allowed us to determine threshold values that would result in a favorable C-E ratio (<$10,000), which would be achieved if the efficacy of ultrasound-guided liver biopsy in preventing major complications were ![]() 60% (Fig. 2). A recent randomized controlled study comparing “blind” liver biopsies to ultrasound-guided liver biopsies reported a 78% reduction in the incidence of major complications in the ultrasonography group.8 Similarly, Papini et al. reported a 75% reduction in complications by the use of ultrasound guidance.5 The additional cost of ultrasonography is probably the most sensitive variable in this analysis. Ultrasonography costs do not add prohibitively to the cost of liver biopsy. Costs can be further lowered at centers in which a large number of liver biopsies are performed. Creating a “liver biopsy unit” staffed by a team of physicians and/or physician assistants trained in ultrasound-guided biopsies may lower operating costs and thus reduce complications at a reasonable cost.
60% (Fig. 2). A recent randomized controlled study comparing “blind” liver biopsies to ultrasound-guided liver biopsies reported a 78% reduction in the incidence of major complications in the ultrasonography group.8 Similarly, Papini et al. reported a 75% reduction in complications by the use of ultrasound guidance.5 The additional cost of ultrasonography is probably the most sensitive variable in this analysis. Ultrasonography costs do not add prohibitively to the cost of liver biopsy. Costs can be further lowered at centers in which a large number of liver biopsies are performed. Creating a “liver biopsy unit” staffed by a team of physicians and/or physician assistants trained in ultrasound-guided biopsies may lower operating costs and thus reduce complications at a reasonable cost.
Major advances in the therapy of liver diseases have led to more frequent use of liver biopsy. Hence, small changes in the technique may result in large changes in its costs and benefits. Recommending ultrasonography for routine liver biopsies will increase costs, and this must be balanced in the face of potential benefits gained from reduction in complications. We performed this analysis with a consistent bias favoring “blind” biopsies and did not include quality adjustments or indirect costs associated with complications, which would strengthen our conclusions. However, the analysis should be interpreted with caution. First, the clinical data supporting the efficacy of ultrasonography are limited. Only one randomized study has compared ultrasonography and “blind” biopsies.8 A second study randomized only one group of patients.5 However, both studies have shown up to a 75% reduction in major complications in the ultrasonography group. Second, the efficacy of ultrasonography demonstrated in randomized studies does not mean its use is “effective”; that is, the observed benefit in randomized studies may not be observed in clinical practice or at other centers. Third, the perspective of the analysis was that of a payer and was not societal. Hence, cost savings estimated by the model relate to savings to the payers. Nevertheless, this exploratory analysis was designed to study the economic impact of performing ultrasound-guided liver biopsies instead of “blind” percutaneous liver biopsies. Sensitivity analysis using a wide range of costs did not change our final conclusions.
In conclusion, our decision analytic model suggests that ultrasound-guided liver biopsy would have a C-E ratio at least comparable with those of other health care interventions when the frequency of major complications after percutaneous liver biopsies is greater than 2% and ultrasonography reduces ![]() 60% of the complications. If the additional cost of ultrasound guidance were
60% of the complications. If the additional cost of ultrasound guidance were ![]() $67, in addition to reducing major complications, potential cost savings could be expected.
$67, in addition to reducing major complications, potential cost savings could be expected.
Footnotes
Abbreviations:
C-E ratio, cost-effectiveness ratio.
Presented at the annual meeting of the American Association for the Study of Liver Diseases, November 1996
Received October 7, 1997; accepted January 26, 1998.
Address reprint requests to: K. D. Lindor, M.D., Mayo Clinic, 200 First Street SW, Rochester, MN 55905. Fax: (507) 284-0538.
Copyright © 1998 by the American Association for the Study of Liver Diseases.